Fungal pathogen-host interactions

Invasion by fungal pathogens is important, as fungal pathogens account for as many deaths each year as tuberculosis or malaria. Candida species are the most common human fungal pathogens, with Candida albicans being the most pathogenic. Its medical importance is highlighted by its recent inclusion in the ‘Critical Priority Group’ of the WHO fungal priority pathogens list (WHO FPPL 2022). This opportunistic pathogen colonizes the skin, genital and intestinal mucosa of most healthy individuals and is part of the normal commensal flora. In susceptible hosts, C. albicans can invade the gastrointestinal mucosa and enter the bloodstream, leading to severe systemic infection.
While invasion of the host epithelium represents the first step of C. albicans infection, this process remains partially understood. A thorough understanding of the molecular and cellular mechanisms underlying C. albicans pathogenicity is essential for the development of future prevention and treatment strategies.
We employ a multi-scale microscopy-based approach to study the invasive lifestyles of C. albicans during in vitro epithelial infection. We use multi-dimensional live cell imaging combined with damage-sensitive reporters to track invasion and damage in epithelial layers in real-time. To obtain additional information, the three-dimensional nano-scale organization of sites of interest is analyzed using advanced ‘volume’ electron microscopy (vEM). Microscopy assays are combined with genetic and cellular perturbations providing functional data.
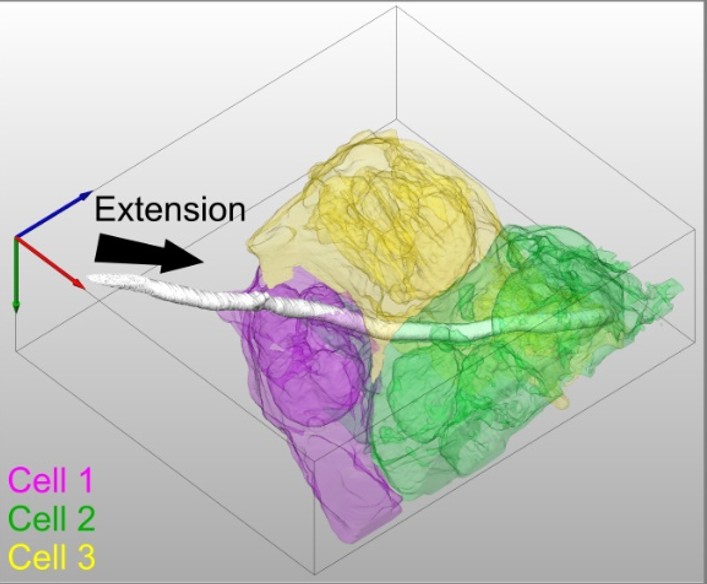
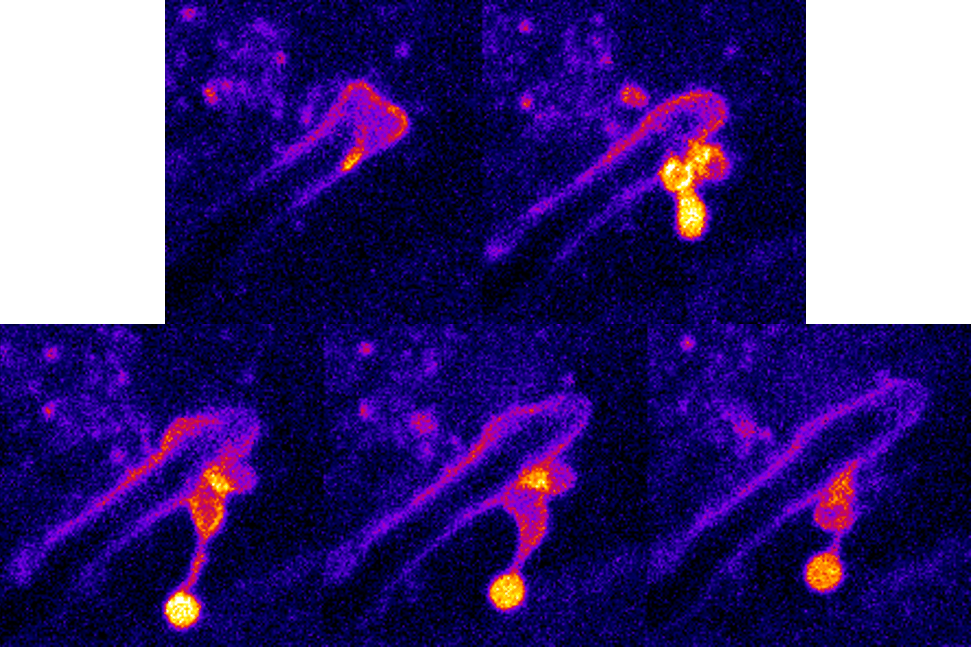
We have recently shown that invasion can result in host membrane breaching occurring at distinct host subcellular niches, or alternatively, in the formation of host membrane-derived trans-cellular tunnels that allow hyphae to traverse several neighboring host cells without host damage. Thus, C. albicans can occupy different invasive niches during infection.
Our goal is to reveal the cellular and molecular determinants and mechanisms underlying C. albicans epithelial infection, and shed light on the first steps of disease caused by this medically important fungal pathogen.
The team members
Our research focus on 3 axes
- Fungal pathogens
- Candida albicans
- Live cell imaging
- Volume electron microscopy
- Pathogen-host interactions
- Invasion
1. Understanding the mechanisms of invasion and damage during C. albicans epithelial invasion
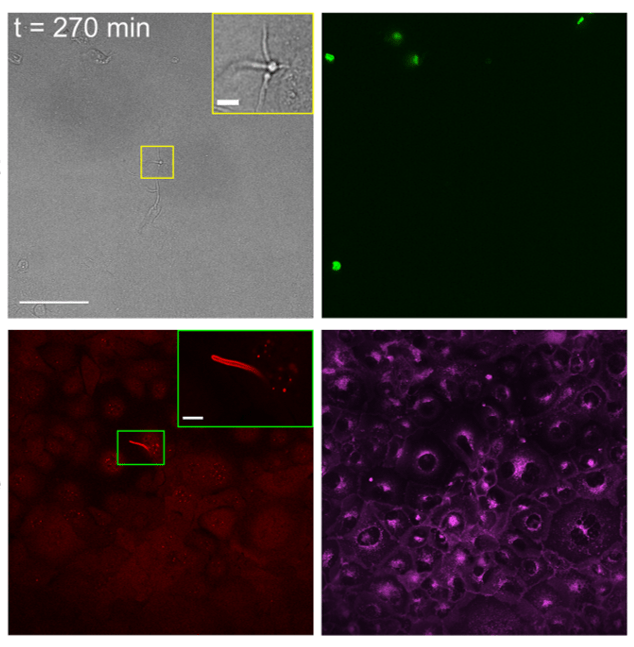
We are employing different cell lines stably expressing damage-sensitive reporters to study invasion and damage by C. albicans hyphae in real-time. Live cell imaging combines different reporters for membrane rupture, host cell death, host membrane staining and others, providing a real-time view of key infection events. Assays are performed in a multi-well format, allowing direct comparison of different experimental conditions, such as WT C. albicans strain vs. strains that do not express key virulence factor. High resolution views of sites of interest are obtained by using serial block face-scanning electron microscopy (SBF-SEM), a type of volume EM, capable of revealing large cellular environments in three-dimensions at nanoscale resolution.
2. Establishment of advanced experimental infection models.
As monolayer epithelial models are limited in their physiological relevance, we aim to develop advanced three-dimensional models to better mimic infection. For this purpose, we are currently establishing a trans-well based stratified epithelium infection model suited for high-resolution stable live cell imaging. Once in place, infection of stratified oral epithelia will be modeled in real-time allowing observation and quantification of infection of a multi-layered tissue using WT and mutant albicans strains as well as other functional perturbations.
3. Exploring infection by other fungal pathogens.
The technical expertise and know-how developed in our group for studying C. albicans infection in vitro is directly transferrable to the study of other fungal pathogens. We have recently begun to explore the infection mechanisms of Candida parapsilosis and Aspergillus fumigatus.
The opportunities
- Provide novel insights into the mechanism of fungal epithelial infection
- Apply cutting-edge microscopy approaches to the study of pathogen-host interactions
- Develop new strategies to combat the emerging threat of fungal pathogens
Publications
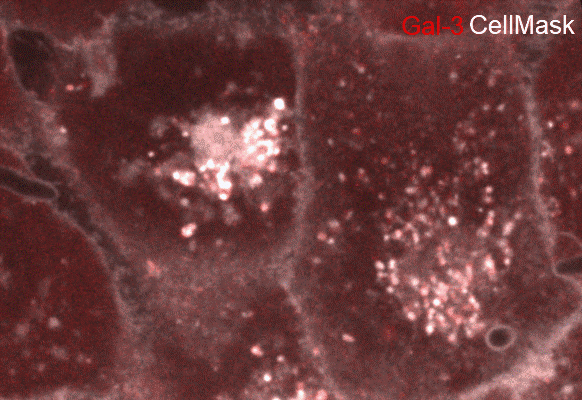
Candida albicans epithelial invasion induces host membrane rupture at distinct subcellular niches following priming by the fungal toxin candidalysin. Conan N., Marthe L., Shekoory A., Latour-Lambert P., Franetich JF., Larsen M., Weiner A. BioRxiv (2025); doi: https://doi.org/10.1101/2025.05.13.653632
The AMA1-RON complex drives Plasmodium sporozoite invasion in the mosquito and mammalian hosts. PLoS Pathog. 2022 Jun 22;18(6):e1010643. doi: 10.1371/journal.ppat.1010643. eCollection 2022 Jun.PMID: 35731833 Free PMC article.
Trans-cellular tunnels induced by the fungal pathogen Candida albicans facilitate invasion through successive epithelial cells without host damage. Nat Commun. 2022 Jun 30;13(1):3781. doi: 10.1038/s41467-022-31237-z. Free PMC article.
Step by step guide to post-acquisition correlation of confocal and FIB/SEM volumes using Amira software. Weiner A. Methods in cell biology: Correlative Light and Electron Microscopy (2020) IV Volume 1622020.
Fundamental helical geometry consolidates the plant photosynthetic membrane. Proc Natl Acad Sci U S A. 2019 Oct 29;116(44):22366-22375. doi: 10.1073/pnas.1905994116.
The Pathogen-Host Interface in Three Dimensions: Correlative FIB/SEM Applications. Trends Microbiol. 2019 May;27(5):426-439. doi:10.1016/j.tim.2018.11.011.
On-site secretory vesicle delivery drives filamentous growth in the fungal pathogen Candida albicans. Cell Microbiol. 2019 Jan;21(1):e12963. doi: 10.1111/cmi.12963.